|
2/14/2024 0 Comments Chemistry reference table
Reference Tables for Physical Setting/Chemistry. 001 milli- m part per million ppm concentration 10Ġ**.** 000 001 micro- (or u) molarity M, mol concentrationġ adapted from: The University of the State of New York. Selected Prefixes degree Celsius ☌ Factor Number of Units Prefix Symbol amt of substance (SI) mol moleġ06 1,000,000 mega- M Joule (SI derived)) Jġ0 −1 0**.** 1 deci- d second (SI) s timeġ0 −2 0**.** 01 centi- c liter L, ℓ volume 10 −3 0. ☏ = degree Fahrenheit mm of mercury mm Hg Torr = millimeter of mercury (mm Hg) gram g NCDPI Reference Tables for Chemistry (2012) Chemistry Reference Tables Name Value Avogadro’s number 6.022 10u 23 particles/mole Gas constant (R) 0.0821 L atm mole K 62.4 L mmHg mole K 8.314 L kPa mole K Standard pressure 1.00 atm 101. “Standard” Pressure 1 atm 760 torr 101**.** 3 kPa meter (SI) m Standard Temperature and Pressure Table B. Some Common & Equivalent Units and Approximate Conversions. Bond Dissociation Energies & Bond Lengths. Ksp Values for Some Insoluble Salts at 25☌. Lu Hf Re Chemistry Reference Tables 1 Table A. Lutetium hafnium tungsten rhenium osmium platinum You are free to share and/or adapt this work for non-commercial purposes, withĪppropriate attribution, under an identical, similar, or compatible license. Attribution-NonCommercial-ShareAlike 4 International License. 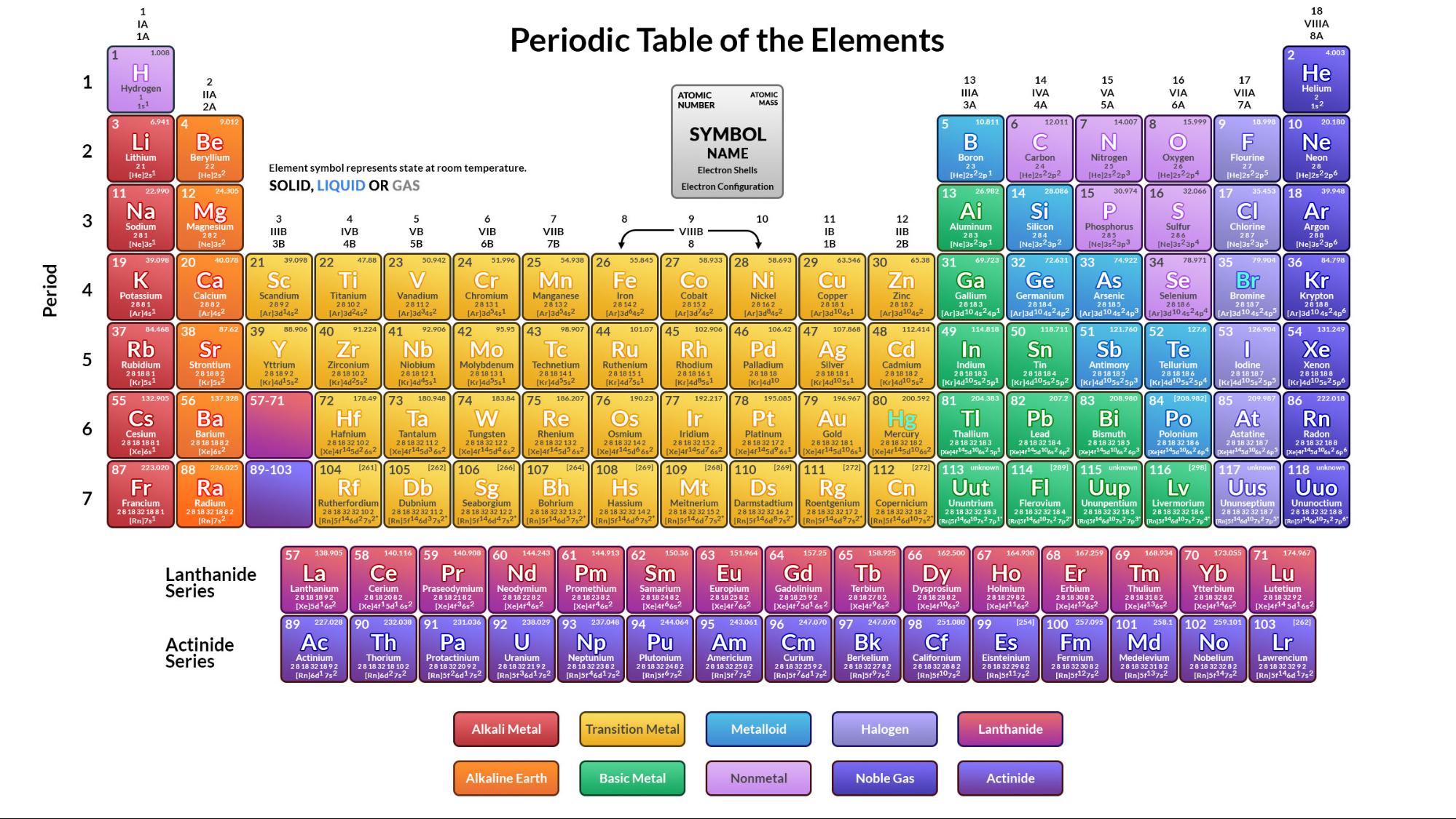
g H 2O, must be used as a conversion factor to calculate the maximum amount of solute, ammonium iodide, NH 4I, that can dissolve in the given amount of solvent, 75.0 grams of water, H 2O.Copyright © 2009–2018 Jeff Bigler. In order to determine whether this solution is saturated or unsaturated, the solubility of ammonium iodide, NH 4I, which has a reported value of 172 g/100. Since the chemical formula for water, H 2O, is associated with the 100.-gram quantities in the denominators of the solubilities in Table 7.9.1, water, H 2O, is the solvent in this solution, and the remaining substance, ammonium iodide, NH 4I, is the solute, "by default." Use the solubility information that is presented in Table 7.9.1 to determine whether the resultant solution is saturated or unsaturated, and calculate the amount of excess solute that remains undissolved in this solution.īefore a solubility limit can be applied as a conversion factor, each substance that is referenced in the given problem must first be classified as a solute or a solvent. 
L, mL liter(s), milliliter(s) mm Hg millimeters of mercury g gram(s) J, kJ joule(s), kilojoule(s) nm nanometer(s) V volt(s). \)Ī solution is prepared by mixing 129 grams of ammonium iodide and 75.0 grams of water at 20 degrees Celsius. ADVANCED PLACEMENT CHEMISTRY EQUATIONS AND CONSTANTS Throughout the test the following symbols have th e definitions specified unless otherwise noted.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
